How COPIKTRA May Help with CLL/SLL
FDA approval for COPIKTRA was based on a study of 196 people with CLL or SLL who had at least two prior therapies.
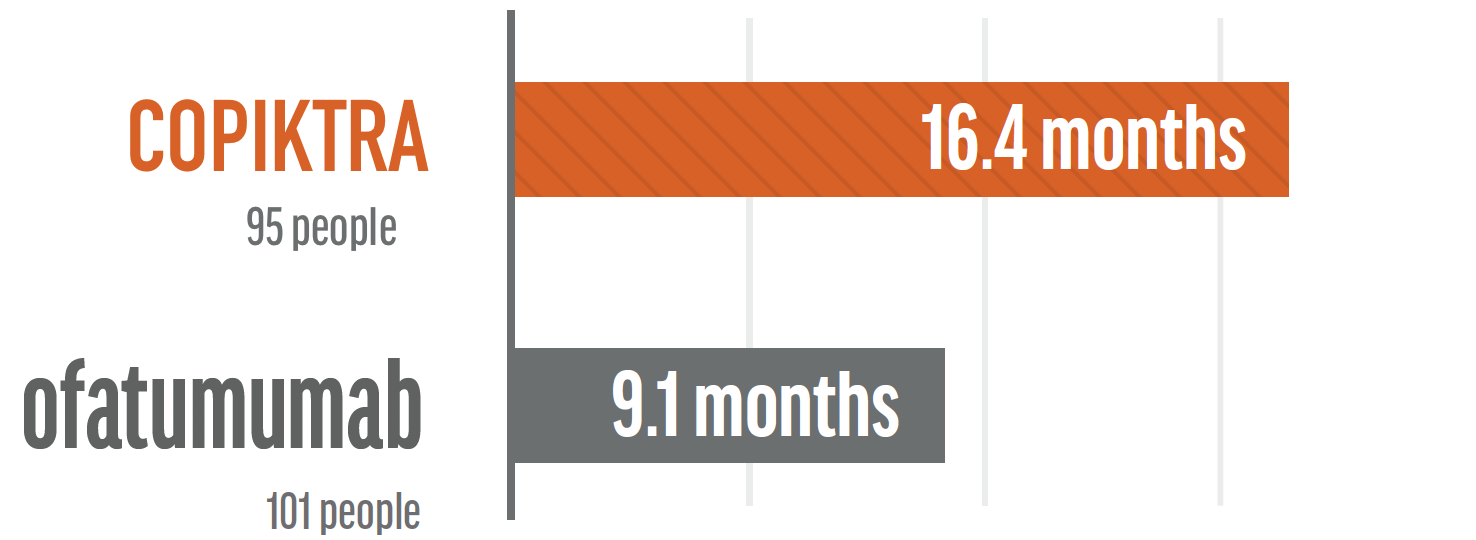
- 95 received COPIKTRA compared with 101 who received ofatumumab
- For those taking COPIKTRA, time without their disease worsening was extended by 7.3 months compared with people who received ofatumumab
People treated with COPIKTRA showed a 60% lower risk of progression compared to those treated with ofatumumab
See study results below.COPIKTRA extended progression-free survival by 7.3 months in patients treated for CLL/SLL when compared to ofatumumab.
- Progression-free survival (PFS) is the length of time on treatment that someone lives with cancer but it does not get worse. The study measured PFS with COPIKTRA compared to ofatumumab.
- For those people in the study, the median progression-free survival was 16.4 months for COPIKTRA vs. 9.1 months for ofatumumab. Median means the middle value, so half of the people in the study would fall above the value and half of the people would fall below the value.
More than 7 out of 10 People with CLL/SLL Responded to Treatment with COPIKTRA
- The study measured overall response rate (ORR) in people who had received at least two prior therapies. ORR is a percentage of patients whose cancer shrinks or disappears after treatment.
- A partial response means that fewer signs of the disease were observed in blood tests, x-rays, CT scans, or bone marrow tests in response to the treatment. All responses were partial responses.
 IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION
- Treatment-related mortality occurred in 15% of COPIKTRA treated patients.
- Fatal and/or serious infections occurred in 31% (4% fatal) of COPIKTRA-treated patients. Monitor for signs and symptoms of infection. Withhold COPIKTRA if infection is suspected.
- Fatal and/or serious diarrhea or colitis occurred in 18% (<1% fatal) of COPIKTRA-treated patients. Monitor for the development of severe diarrhea or colitis. Withhold COPIKTRA.
- Fatal and/or serious cutaneous reactions occurred in 5% (<1% fatal) of COPIKTRA-treated patients. Withhold COPIKTRA.
- Fatal and/or serious pneumonitis occurred in 5% (<1% fatal) of COPIKTRA-treated patients. Monitor for pulmonary symptoms and interstitial infiltrates. Withhold COPIKTRA.
WARNINGS AND PRECAUTIONS
Treatment-related Mortality: In a randomized controlled study in patients with relapsed or refractory CLL or SLL, treatment with COPIKTRA caused increased treatment-related mortality. With extended follow-up with a median of 63 months, treatment-related deaths occurred in 15% (23/158) of those patients in the overall population. In the indicated patient population, patients with relapsed or refractory CLL or SLL after at least two prior lines of systemic therapy, treatment related deaths following treatment with COPIKTRA occurred in 14% (13/93) of patients. The most common cause of the treatment-related deaths were infections, which occurred in 9% and 11% of patients with relapsed or refractory CLL following at least one or two prior systemic therapies, respectively. COPIKTRA is not indicated and is not recommended for any patients in the initial or second-line treatment setting.
Infections: Serious, including fatal (18/442, 4%), infections occurred in 31% of patients receiving COPIKTRA. The most common serious infections were pneumonia, sepsis, and lower respiratory infections. Median time to onset of any grade infection was 3 months, with 75% of cases occurring within 6 months. Treat infections prior to initiation of COPIKTRA. Advise patients to report new or worsening signs and symptoms of infection. Cases of Pneumocystis jirovecii pneumonia (PJP) (1%) and cytomegalovirus (CMV) reactivation/infection (1%) occurred in patients taking COPIKTRA. Provide prophylaxis for PJP during treatment and following completion of treatment until the absolute CD4+ T cell count is greater than 200 cells/μL. Consider prophylactic antivirals during COPIKTRA treatment to prevent CMV infection including CMV reactivation.
Diarrhea or Colitis: Serious, including fatal (1/442; 0.2%), diarrhea or colitis occurred in 18% of patients receiving COPIKTRA. Median time to onset of any grade diarrhea or colitis was 4 months, with 75% of cases occurring by 8 months. The median event duration was 0.5 months. Advise patients to report any new or worsening diarrhea.
Cutaneous Reactions: Serious, including fatal (2/442; 0.5%), cutaneous reactions occurred in 5% of patients receiving COPIKTRA. Fatal cases included drug reaction with eosinophilia and systemic symptoms (DRESS) and toxic epidermal necrolysis (TEN). Median time to onset of any grade cutaneous reaction was 3 months with a median event duration of 1 month. Presenting features for the serious events were primarily described as pruritic, erythematous, or maculo-papular. Less common presenting features include exanthem, desquamation, erythroderma, skin exfoliation, keratinocyte necrosis, and papular rash. Advise patients to report new or worsening cutaneous reactions.
Pneumonitis: Serious, including fatal (1/442; 0.2%), pneumonitis without an apparent infectious cause occurred in 5% of patients receiving COPIKTRA. Median time to onset of any grade pneumonitis was 4 months with 75% of cases occurring within 9 months. The median event duration was 1 month with 75% of cases resolving by 2 months.
Hepatotoxicity: Grade 3 and 4 ALT and/or AST elevation developed in 8% and 2%, respectively, of patients receiving COPIKTRA (N=442). Two percent of patients had both an ALT or AST > 3 X ULN and total bilirubin > 2 X ULN. Median time to onset of any grade transaminase elevation was 2 months with a median event duration of 1 month. Monitor hepatic function during treatment with COPIKTRA.
Neutropenia: Grade 3 or 4 neutropenia occurred in 42% of patients receiving COPIKTRA (N=442), with Grade 4 neutropenia occurring in 24% of all patients. Median time to onset of grade ≥3 neutropenia was 2 months. Monitor neutrophil counts at least every 2 weeks for the first 2 months of COPIKTRA therapy, and at least weekly in patients with neutrophil counts < 1.0 Gi/L (Grade 3-4).
Embryo-Fetal Toxicity: COPIKTRA can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus and conduct pregnancy testing before initiating COPIKTRA treatment. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment and for 1 month after the last dose.
ADVERSE REACTIONS
B-cell Malignancies Summary
Fatal adverse reactions within 30 days of the last dose occurred in 8% (36/442) of patients treated with COPIKTRA 25 mg BID. Serious adverse reactions were reported in 289 patients (65%). The most frequent serious adverse reactions that occurred were infection (31%), diarrhea or colitis (18%), pneumonia (17%), rash (5%), and pneumonitis (5%). The most common adverse reactions (reported in ≥20% of patients) were diarrhea or colitis, neutropenia, rash, fatigue, pyrexia, cough, nausea, upper respiratory infection, pneumonia, musculoskeletal pain and anemia.
CLL/SLL
Fatal adverse reactions within 30 days of the last dose occurred in 12% (19/158) of patients treated with COPIKTRA and in 4% (7/155) of patients treated with ofatumumab. Serious adverse reactions were reported in 73% (115/158) of patients treated with COPIKTRA and most often involved infection (38%; 60/158) and diarrhea or colitis (23%; 36/158). The most common adverse reactions with COPIKTRA (reported in ≥20% of patients) were diarrhea or colitis, neutropenia, pyrexia, upper respiratory tract infection, pneumonia, rash, fatigue, nausea, anemia and cough.
For specific information on the management of the adverse reactions above, please review Dose Modifications for Adverse Reactions within the full Prescribing Information.
DRUG INTERACTIONS
CYP3A4 Inducers: Coadministration with a strong or moderate CYP3A4 inducer may reduce COPIKTRA efficacy. Avoid coadministration with strong or moderate CYP3A4 inducers.
CYP3A4 Inhibitors: Coadministration with a strong CYP3A4 inhibitor may increase the risk of COPIKTRA toxicities. Reduce COPIKTRA dose when coadministered with a strong CYP3A4 inhibitor.
CYP3A4 Substrates: Coadministration of COPIKTRA with sensitive CYP3A4 substrates may increase the risk of toxicities of these drugs. Consider reducing the dose of the sensitive CYP3A4 substrate and monitor for signs of toxicities of the coadministered sensitive CYP3A4 substrate.
Please see accompanying full Prescribing Information, including Boxed Warning.
To report Adverse Reactions during use of COPIKTRA, contact Secura Bio, call 1-844-9Secura or 1-844-973-2872 and/or to FDA at www.fda.gov/medwatch or call 1-800-FDA-1088.
USCPR2419101



 RESIZE
RESIZE HEALTHCARE PROVIDERS
HEALTHCARE PROVIDERS